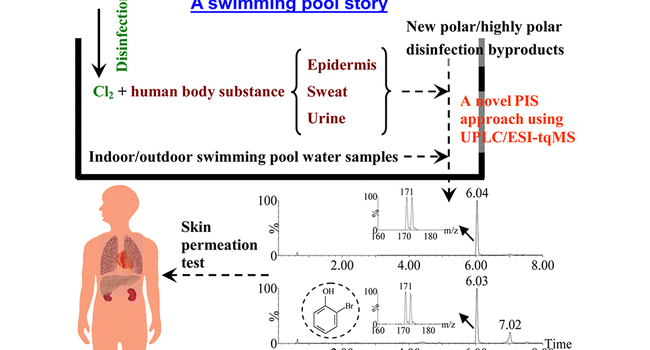
Byproducts in Swimming Pool Water.
Chlorine is widely used for disinfecting public swimming pool water. The disinfectant chlorine, protecting swimmers from pathogenic infection in swimming, may be responsible for some adverse effects on swimmers’ skin and health. In this study, numerous new halogenated disinfection byproducts (DBPs) in chlorinated pool water were detected with a powerful precursor ion scan method using electrospray ionization triple quadrupole mass spectrometry, with or without preseparation with ultra performance liquid chromatography. These new pool DBPs were demonstrated to be mainly halo(nitro)phenols, resulting from chlorination of human body substances (such as urine) in the presence of bromide. Among these new DBPs, 2,4-dibromophenol, 2,4-dichlorophenol, 2-bromophenol, 2,6-dibromo-4-nitrophenol, 2-bromo-6-chloro-4-nitrophenol, and 2,6-dichloro-4-nitrophenol were fully identified or confirmed. For 2,4-dibromophenol, 2,4-dichlorophenol and 2-bromophenol with pure standard compounds available, their permeability values across human skin were measured to be 0.031, 0.021, and 0.023 cm/h, respectively. The effects of chlorine on human skin were also investigated. The interaction of chlorine with epidermis was found to generate many new halogenated DBPs as well as common DBPs; the corneous layer was observed to become rough and even form larger pores after chlorine interaction. It is recommended that swimmers should avoid urinating in pools, and avoid prolonged swimming to reduce chlorine contact and prevent accelerated permeation of DBPs across skin.